Introducing Giga Binding Units (gBU): A Functional Measurement Standard for Immunoglobulin Ingredients

Walk into any supplement retailer or browse online marketplaces for immunoglobulin products, particularly IgG and IgY, and you will notice something striking. Products making similar digestive and immune health claims recommend vastly different daily servings; one product suggests 200mg, another suggests 2,500mg, over 12 times higher. This variation makes it difficult for consumers to evaluate product value, for formulators to select ingredients and establish serving sizes, and for researchers to compare outcomes across studies. For a category valued at more than $5 billion and growing at a CAGR of more than 7%a, this is a challenge worth solving1-3.
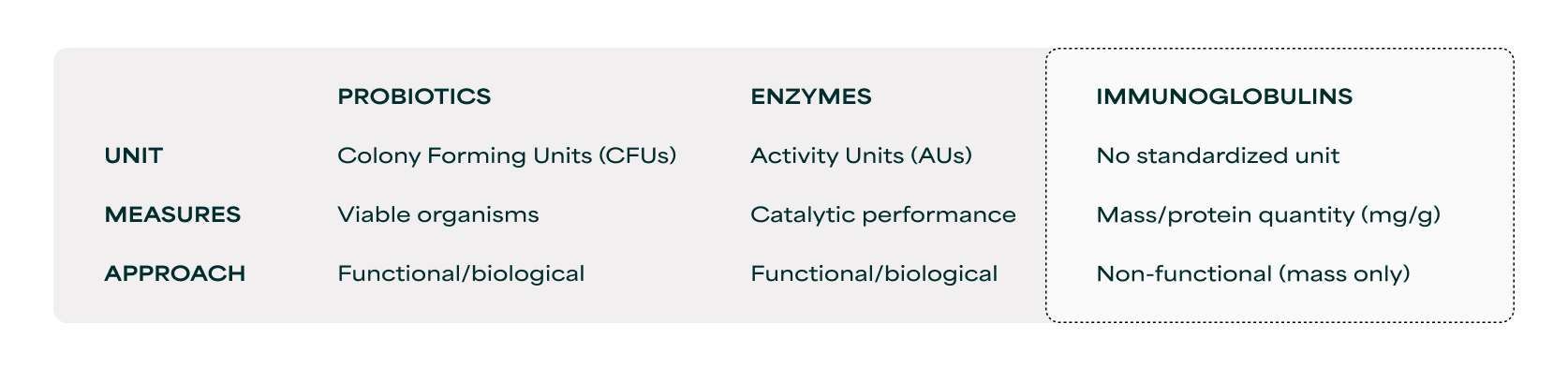
Other functional ingredient categories have faced similar measurement challenges and solved them by standardizing around the biological function by which they deliver health benefits, rather than mass. Probiotics use colony forming units (CFUs) to quantify viable organisms rather than bacterial mass. Enzymes use activity units (AUs) to quantify catalytic performance, rather than protein quantity. Immunoglobulins require the same approach, yet today the category relies solely on mass-based measurement with no standard that captures the functional benefits these ingredients deliver.
Measurement standards across functional ingredients
To see why functional measurement matters, we need to understand how immunoglobulins deliver health benefits as nutraceutical ingredients. The biological function of immunoglobulins in nutraceuticals derives from their ability to bind specifically to molecular targets in the gut lumen and physically block those targets from interacting with gut cells or other molecules. This binding ability is how immunoglobulins were first discovered in 1890 by Emil von Behring, who called them "antitoxins" for neutralizing bacterial toxins. Today, it remains the primary mechanism through which immunoglobulins in mother's milk protect the newborn baby.
a Combined market estimate spanning colostrum, bovine plasma derivatives, and egg yolk immunoglobulins.
- These statements have not been evaluated by the Food and Drug Administration. These products are not intended to diagnose, treat, cure, or prevent any disease.
This document contains scientific information for business-to-business use and medical professionals, and is not intended as healthcare advise for consumers.
How do Immunoglobulins work?
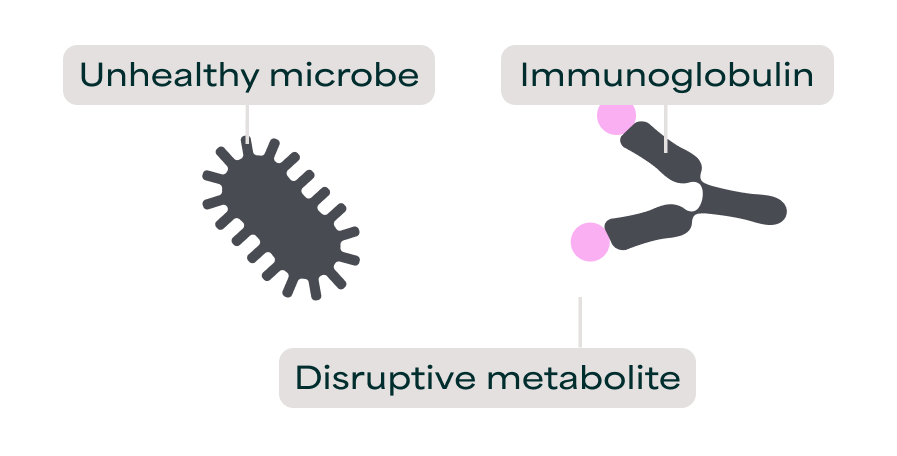
It is the conserved molecular structure of immunoglobulins that determines their biological function. All immunoglobulins have a Y-shaped structure with one binding site at the tip of each arm, providing two binding sites per molecule, each capable of binding the same molecular target. This architecture remains consistent across animal species and immunoglobulin classes.
The most common commercial immunoglobulin classes are Immunoglobulin G (IgG) from bovine sources and Immunoglobulin Y (IgY) from chicken sources, both consisting of a single Y-shaped unit with two binding sites per molecule.
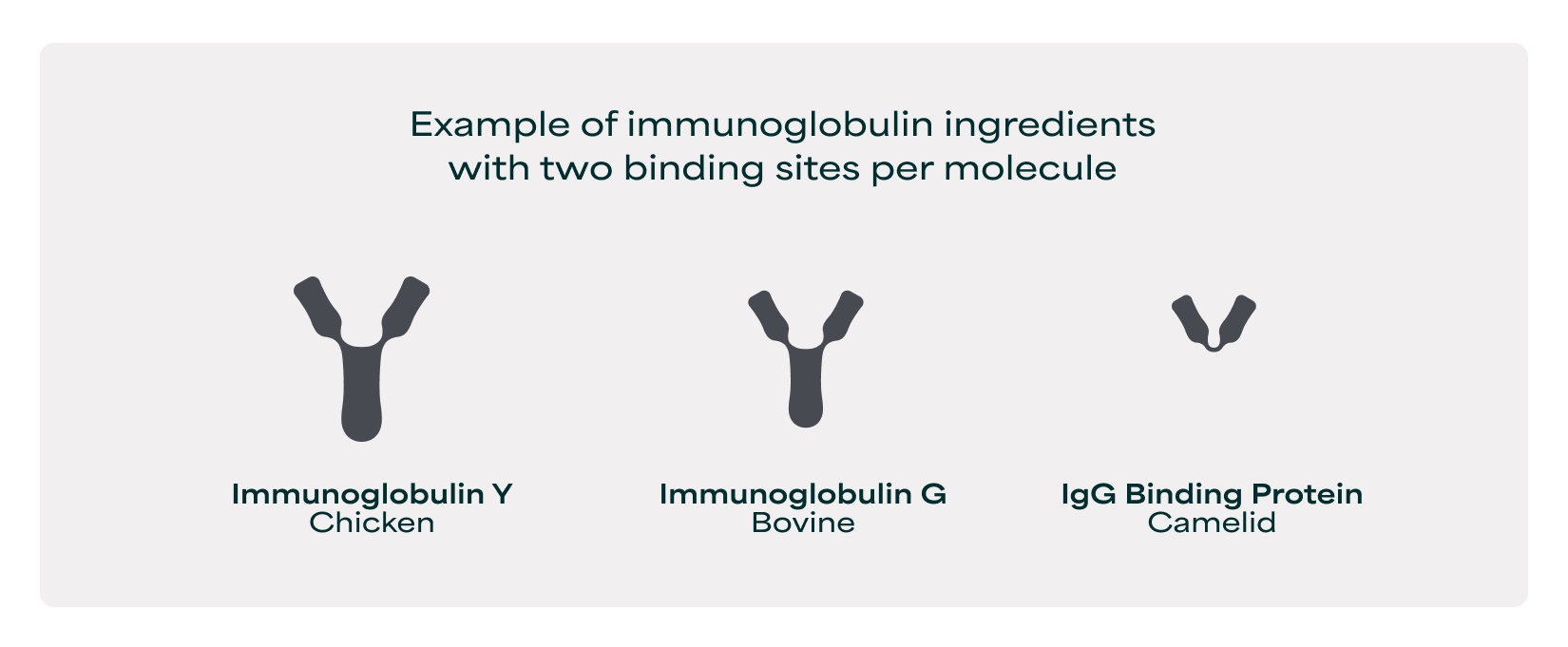
However, some immunoglobulin classes form assemblies where multiple Y-shaped units combine, such as Immunoglobulin M (IgM), which arranges five identical immunoglobulins into a pentamer with a capacity of ten total binding sites.
Another example, specific to the gut, is the organization of the secretory IgA (sIgA) molecule, where two immunoglobulin A molecules are joined by a J-chain which protects the combined protein structure in the gut lumen – with a total 4 binding sites combined for one sIgA. This structural conservation makes binding sites quantifiable and comparable, where a greater number of binding sites means greater ability to deliver health benefits associated with microbiota and immune function.
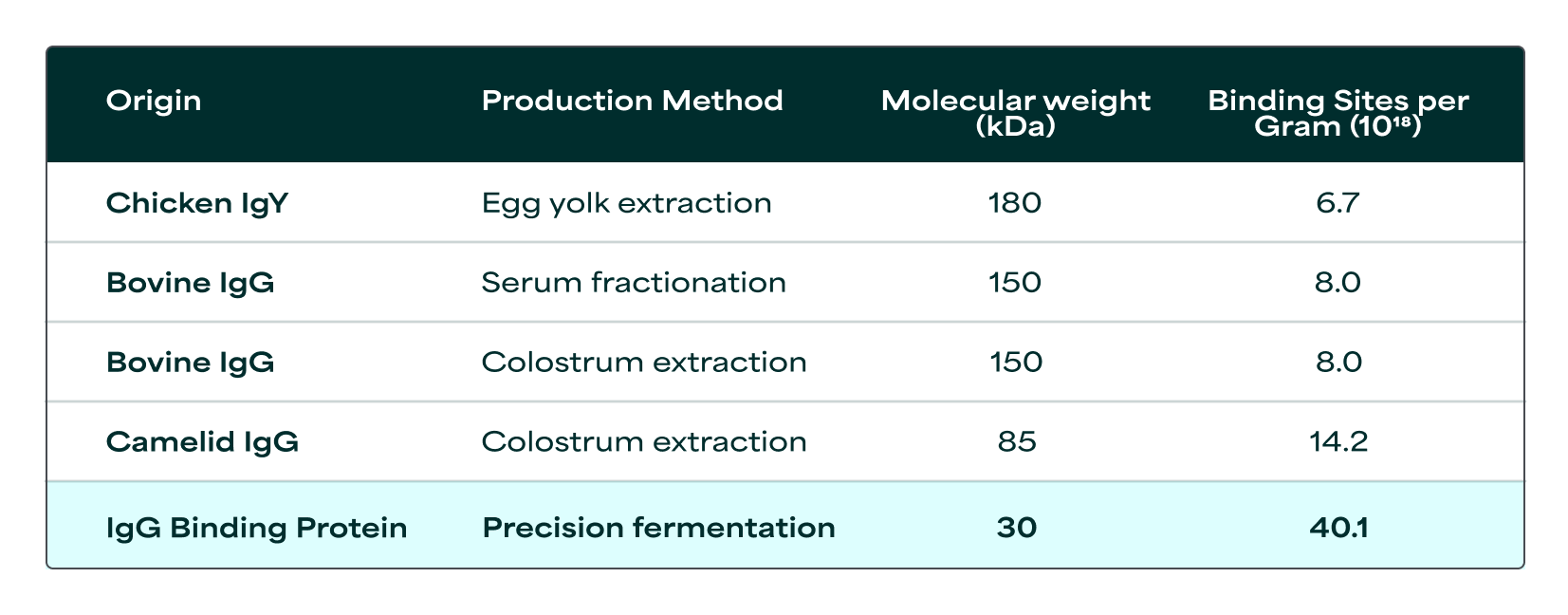
A five-fold variation in binding site density across Immunoglobulin source
Commercially available immunoglobulin ingredients are derived from bovine sources (colostrum, serum, plasma), chicken sources (egg yolk), and camelid sources (colostrum or precision fermentation). These sources yield immunoglobulins with distinct molecular structures and weights. This results in binding site densities that vary more than 5-fold across sources (Table 1). This 5-fold variation means that 100mg of one source could deliver the same amount of binding sites, and thereby functionality, as 500mg of another. This is valuable information that mass-based labeling alone does not capture. Furthermore, production methods affect ingredient purity, so the actual immunoglobulin content per gram of finished product varies even when the immunoglobulin origin remains constant. A functional measurement standard would make these differences visible, enabling comparison based on capacity to deliver health benefits.
Giga Binding Units (gBU): A new functional measurement standard
Giga Binding Units (gBU) are a new approach to quantifying binding sites per serving, expressed in billions for practical relevance. It measures the biological mechanism through which immunoglobulin ingredients deliver health benefits rather than pure mass, for transparent communication of product potency. Furthermore, gBU solves the purity problem inherent in mass-based measurement by accounting for the actual immunoglobulin content in the finished ingredients by quantifying binding sites per daily serving.
A product with 50% immunoglobulin purity and another with 90% purity become directly comparable, just as probiotic products with varying formulation and formatting become comparable through CFU measurement. The gBU measurement system applies universally across all immunoglobulin sources and production methods, from traditional bovine colostrum extraction to precision-fermented camelid fragments. Any manufacturer or formulator can calculate gBU using standard certificate of analysis data, immunoglobulin content, and molecular composition.
Calculating gBU requires information readily available from standard certificates of analysis, including the total immunoglobulin content, the immunoglobulin class composition, and the molecular weight of each class present. The calculation follows directly from the structural principles established above. Each Y-shaped immunoglobulin unit contains two binding sites. Single-unit classes like IgG and IgY provide two binding sites per molecule, while multi-unit assemblies like sIgA provide four binding sites and IgM provides ten binding sites per molecule. Using the serving size, purity, molecular weight, and binding sites per molecule, the total gBU per serving can be determined.
An online gBU calculator is available at https://gbu-calculator.bindingproteins.com to simplify this calculation for any immunoglobulin product, with the detailed mathematical formula provided in Appendix A.
When applied to commercial immunoglobulin products, gBU reveals functional equivalencies obscured by mass-based measurement (Table 2). Products with vastly different serving recommendations become directly comparable when expressed in gBU. A 200 mg serving may deliver equivalent gBU to a 1,200 mg serving from a different source, revealing that the six-fold mass difference reflects molecular structure rather than functional capacity. This transparency enables consumers to evaluate value based on functional delivery, allows formulators to optimize serving sizes across ingredient sources, and provides researchers with standardized metrics for comparing outcomes across studies using different immunoglobulin types.

The gBU measurement standard enables functional comparison across stakeholders in the value chain
- For consumers, gBU allows purchasing decisions based on binding sites rather than arbitrary dosage numbers.
- For formulators, gBU enables objective ingredient comparison and cost optimization per functional unit across different sources.
- For researchers, gBU provides the standardized metric necessary to compare dosages across studies using different immunoglobulin sources, enabling the cumulative evidence building essential for regulatory recognition and health claim substantiation.
- For the category broadly, gBU creates competitive transparency where all manufacturers can demonstrate the functional value their ingredients deliver, whether through traditional extraction methods or newer production approaches. This follows the standardization path established by probiotics, vitamins, and enzymes.
Future refinement of the gBU metric can incorporate target specificity, indicating not only the amount of binding sites but also what molecular targets the immunoglobulins bind. This would parallel probiotic measurement evolution, where CFU established the baseline and strain-level specification followed. Probiotics transitioned from confusion to CFU standardization through scientific consensus, industry collaboration, and regulatory recognition. The immunoglobulin category stands at a similar inflection point. Widespread adoption requires ingredient manufacturers to specify binding site capacity on certificates of analysis, formulators to incorporate gBU into serving size decisions, researchers to report gBU alongside mass in studies for claim substantiation, and industry organizations to establish measurement best practices.
By measuring binding sites rather than mass, the immunoglobulin category can establish the transparent, function-based standardization necessary for consumer transparency and adoption.
Appendix
Appendix A - gBU Calculation Methodology
The gBU formula calculates total binding sites per serving expressed in billions.
Formula
gBU = [Serving size in grams × Immunoglobulin purity × Binding sites per molecule × 6.022 × 1023] ÷ [Molecular weight in daltons × 109]
Where each variable represents
- Serving size in grams = the recommended daily dose converted to grams
- Immunoglobulin purity = the percentage of immunoglobulin in the ingredient expressed as a decimal
- Binding sites per molecule = 2 for IgG and IgY, 4 for secretory IgA, 10 for IgM
- 6.022 × 1023 = Avogadro's number
- Molecular weight in daltons = the molecular weight of the immunoglobulin class
- 109 = conversion factor to express result in billions
Worked Example
A bovine colostrum ingredient containing 50% IgG with molecular weight 150,000 Da in a 500mg serving.
Convert serving size to grams = 500mg ÷ 1,000 = 0.5g
Express purity as decimal = 50% = 0.50
Binding sites per molecule for IgG = 2
Calculate:
[0.5 × 0.50 × 2 × 6.022 × 1023] ÷ [150,000 × 109] = 2.0 gBU
This ingredient delivers 2.0 gBU per 500mg serving.
References
- Precedence Research. Colostrum Market Size. 2024.
- Precedence Research. Bovine Blood Plasma Derivatives Market. 2024.
- Research Intelo. Egg Yolk IgY Market. 2024.
These statements have not been evaluated by the Food and Drug Administration. These products are not intended to diagnose, treat, cure, or prevent any disease.
This document contains scientific information for business-to-business use and medical professionals, and is not intended as healthcare advise for consumers.


.png)