Gut Defense: How IgG Binding Protein LT Supports Barrier Integrity Over Conventional Immunoglobulins

Why gut-health solutions matter: The importance of a balanced microbiota
In the last decade, gut health has rapidly gained importance among consumers and researchers, driven by increased awareness and advancing scientific evidence. Central to this interest is mounting research showing that a balanced gut microbiota supports overall health, while an unbalanced gut microbiota is linked to metabolic, immune, and gastrointestinal (GI) disorders.1,2 Together with the intestinal barrier, the gut microbiota forms a critical defense system protecting the body against unwanted bacteria.
However, this defense system is sensitive and can easily be disturbed by external factors like antibiotics, poor diet, and unhealthy metabolites, leading to gut dysbiosis.3 Gut dysbiosis with its reduced microbial diversity, can cause a person to be more susceptible to GI concerns, highlighting the need for solutions that support microbial resilience.4 This knowledge has led to a variety of gut health solutions that preserve or restore gut health balance, with a particular focus on supporting the gut microbiota’s core functions: digestion, metabolism, and immune regulation.
Immunoglobulin-based approaches to gut resilience
Immunoglobulins are essential for the body’s immune response and serve as key components supporting gut resilience.5,6 Maternal immunoglobulins in breast milk exemplify this by providing passive immune support and protecting infants during the critical early period.7 There are currently various immunoglobulin-containing gut health supplements on the market, including bovine colostrum (BC), serum-derived bovine immunoglobulin isolate (SBI), and immunized egg powder containing the avian immunoglobulin IgY. These products are intended to promote microbial diversity, bind unwanted bacteria, support gut barrier integrity, and modulate local immune responses.8–10 By promoting the body’s natural immune defense, they help maintain a balanced gut microbiota, supporting overall digestive wellness. Although BC is by far the most well-known and studied immunoglobulin supplement, SBI and immunized egg powder have gained attention in recent years for their more concentrated immunoglobulin content and as dairy-free colostrum alternatives.

The next step in gut health products – Enter IgG Binding Proteins
IgG Binding Proteins represent a novel class of functional protein ingredients for supporting gut health. The ingredients are derived from the binding domain of camelid immunoglobulins, making them an ideal alternative to conventional immunoglobulin products.16 Furthermore, IgG Binding Proteins are produced via precision fermentation, ensuring consistent quality without the use of animals in production. Where conventional immunoglobulin supplements promote the microbiota’s capacity to resist threats, IgG Binding Proteins act directly on unwanted metabolites, offering targeted, consistent, and customizable support. They are documented to bind unwanted metabolites, enabling their safe passage through the gastrointestinal tract,17–19 thus avoiding downstream gut effects such as inflammation and degradation of the gut barrier, and helping to support gut stability in a gentle and effective manner. The ingredient offers a favorable safety profile for its intended uses in food and beverages, supported by both toxicity study and in silico allergenicity assessment, together which have demonstrated the safety of consuming IgG Binding Proteins.20
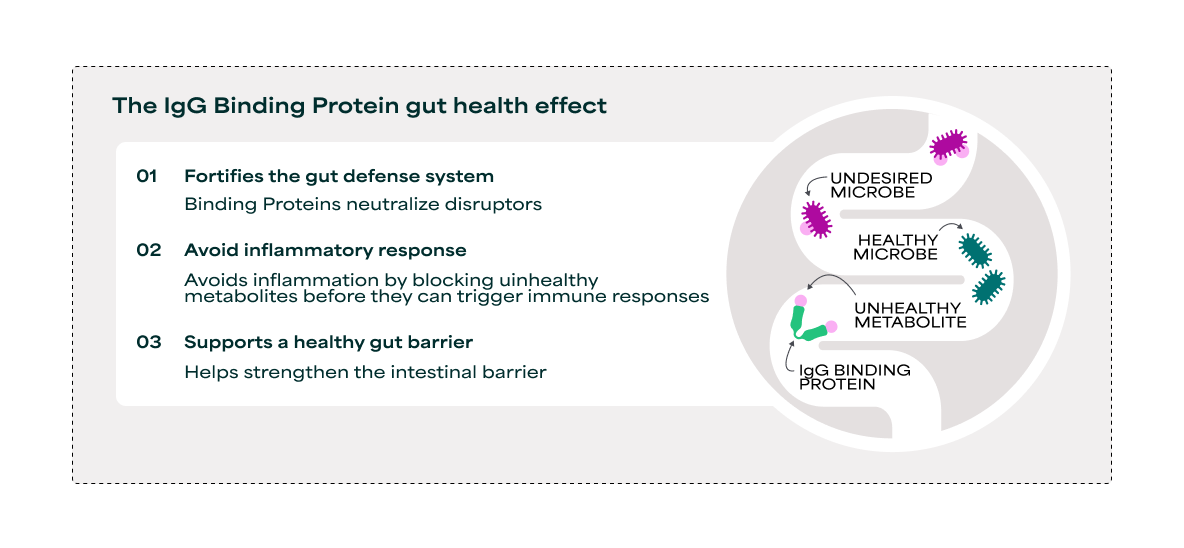
Investigating the power IgG Binding Proteins
Undesired microbes can release unhealthy metabolites in the gut, which disrupts normal gut function and cause fluid loss, local inflammation, and weakening of the gut barrier. As an example, LT toxin from enterotoxigenic E. coli (ETEC), the most common cause of traveler’s diarrhea, can trigger intestinal cells to release fluids and electrolytes.21,22 The fluid loss disrupts the gut barrier, especially tight junctions (the ‘glue’ between cells), causing increased gut permeability and release of inflammatory signals such as interleukin-8 (IL-8). Unlike lipopolysaccharide (LPS), a bacterial cell wall component found on Gram-negative bacteria and present both in the healthy microbiota and unwanted bacteria,23 LT toxin is ETEC-specific. As a result, the toxin can be directly targeted and blocked without compromising a healthy, diverse microbiota.
In order to investigate the ability of different immunoglobulin supplements to strengthen the gut barrier against unhealthy metabolites such as LT toxin from ETEC, in vitro studies have been conducted, including two BC products (Referred to as BC1 and BC2), one SBI product, one IgY product and the novel food ingredient IgG Binding Protein LT. In these studies, three markers were used to examine cells in distress: cyclic adenosine monophosphate (cAMP), IL-8 and tight junctions.
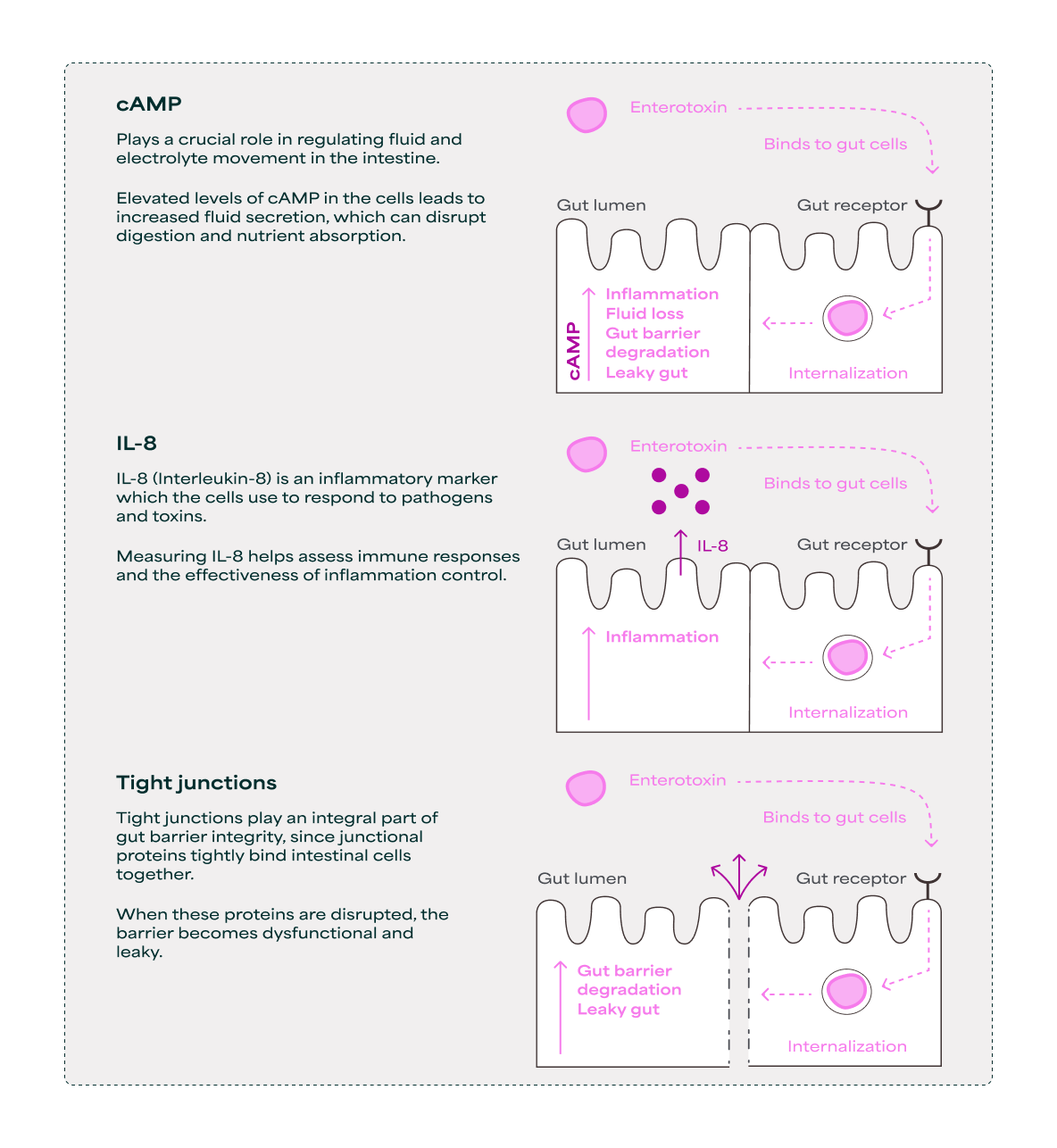
Experimental setup
The most optimal concentration of each product was determined using a cell proliferation assay as according to Playford et al. 2020.24 To simulate the intestinal cell layer two human colonic cell lines were utilized: HCA-7 for measuring cell conversion of ATP to cAMP, and T84 fo rinvestigating inflammation and cell barrier disruption. For the cAMP assay, combinations of toxin (0.2 µg/ml) with different immunoglobulin supplements were added to cells seeded 24 hours prior. After 2-hour incubation the cells were lysed and cAMP levels measured. For the IL-8 assay, cells were seeded 14-18 days prior to experiment, to obtain a confluent, differentiated monolayer representing the intestinal barrier. Combinations of toxin (0.1 µg/ml) with different immunoglobulin supplements were added to cells and incubated together for a day. Afterwards, IL-8 concentration was quantified in the cell supernatant using ELISA. To visually inspect the tight junction distribution after treatment, the cells were fixed and stained with Phalloidin.
IgG Binding Protein LT fortifies the gut defense system
IgG Binding Protein LT is documented to neutralize disrupters such as LT toxin from being absorbed by intestinal cells.19 In an in vitro cell model, IgG Binding Protein LT could block LT toxin uptake, whereas the broad immunoglobulin supplements showed no inhibition.
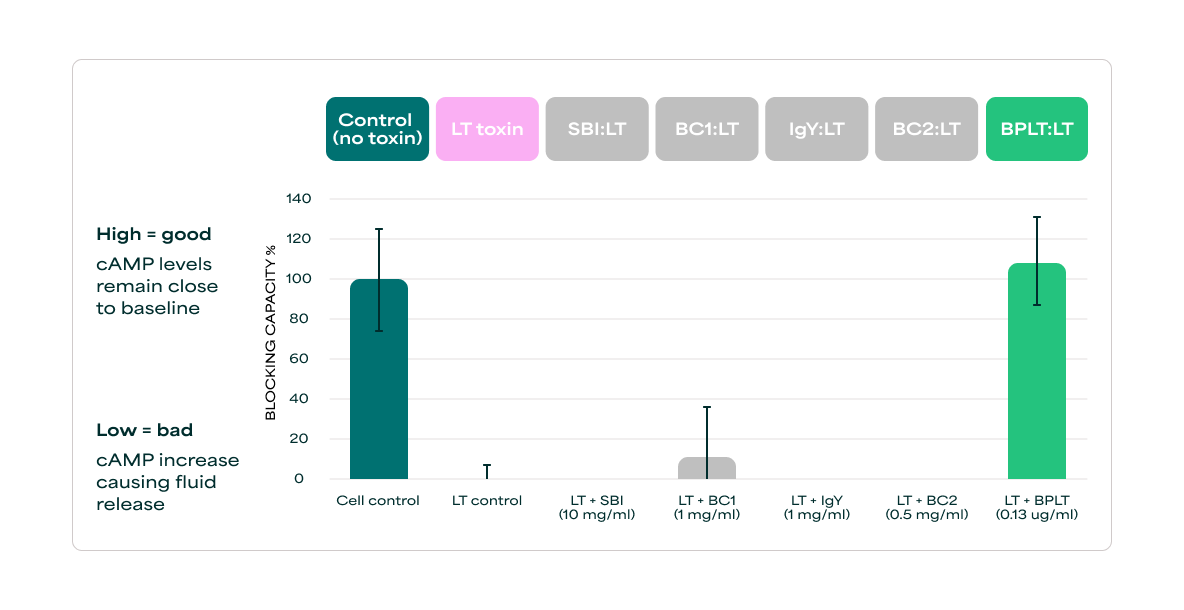
IgG Binding Protein LT avoid inflammatory responses
When the gut barrier integrity is compromised, tight junctions loosen and the epithelium becomes more permeable, triggering an immune response (e.g. release of immune signaling molecules called cytokines). This can drive local inflammation and further cause impairment on the gut barrier. In an in vitro intestinal cell model, IgG Binding Protein LT blocked LT toxin uptake, and no increase in inflammatory signals was observed when cells were exposed to both LT toxin and IgG Binding Protein LT together. The tested immunoglobulin supplements did not reduce inflammation in the presence of LT toxin.

IgG Binding Protein LT supports a healthy gut barrier
The gut barrier helps prevent unhealthy substances from entering the bloodstream while allowing essential nutrients to be absorbed. By blocking LT toxin, IgG Binding Protein LT contributes to the gut defense system by helping maintain the strength and integrity of the gut barrier.

Conclusion
IgG Binding Proteins present a novel food category offering targeted, low-dose, vegan options for supporting gut health in at-risk consumers. This includes travelers, people experiencing occasional GI discomfort, and immune challenged individuals. The novel ingredient is backed by strong safety data and can be integrated in various products such as dietary supplements, functional beverages and dairy products.
The study presented here illustrates how IgG Binding Protein LT protects the intestinal cell barrier from disruption and reduce immune activation by neutralizing the unhealthy metabolite LT toxin from ETEC. Even in low amounts IgG Binding Protein LT demonstrated superior ability to support gut barrier integrity while lowering inflammation in the presence of LT toxin.
In conclusion, IgG Binding Protein LT has the potential to benefit overall gut health, either alone or alongside existing gut support strategies, by helping maintain a strong intestinal barrier and protecting the cells from undesired metabolites in at-risk situations.
References
- Stolfi, C. et al. (2022) Biomedicines 10.
- Fan, Y. & Pedersen, O. (2021) Nat Rev Microbiol 19.
- Hrncir, T. (2022) Microorganisms 10.
- Larsen, O.F.A. & Claassen, E. (2018) Sci Rep 8.
- Cerutti, A. & Rescigno, M. (2008) Immunity 28.
- Jones, E.A. (1972) Gut 13.
- Rogier, E.W. et al. (2014) Proc Natl Acad Sci USA 111.
- Ulfman, L.H. et al. (2018) Front Nutr 5.
- Petschow, B.W. et al. (2014) Clin Exp Gastroenterol 7.
- Han, S. et al. (2021) Front Cell Infect Microbiol 11.
- Playford, R.J. & Weiser, M.J. (2021) Nutrients 13.
- Puppel, K. et al. (2019) Animals 9.
- Müller, S. et al. (2015) Nutr J 14.
- Dumoulin, M. et al. (2002) Protein Sci 11.
- Rathe, M. et al. (2014) Nutr Rev 72.
- Petersson, M. et al. (2023) Trends Biotechnol 41.
- Jenkins, T.P. et al. (2024) npj Biofilms Microbiomes 10.
- Rodriguez Rodriguez, E.R. et al. (2024) Protein Sci 33.
- Petersson, M. et al. (2026) Gut Microbes 18.
- Phipps, K.R. et al. (2025) J Appl Toxicol 45.
- Mudrak, B. & Kuehn, M.J. (2010) Toxins 2.
- Zhang, Y. et al. (2022) Gut Microbes 14.
- Wassenaar, T.M. & Zimmermann, K. (2018) Eur J Microbiol Immunol 8.
- Playford, R.J. et al. (2020) PLoS One 15.
These statements have not been evaluated by the Food and Drug Administration. These products are not intended to diagnose, treat, cure, or prevent any disease.
This document contains scientific information for business-to-business use and medical professionals, and is not intended as healthcare advise for consumers.


.png)